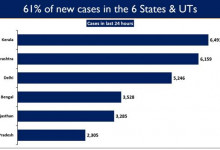
Health
- United States

Xlear Submits COVID-19 Pre-Emergency Use Authorization Request with FDA Regarding Use of Xlear Nasal Spray to Help in Combating SARS-CoV-2
Washington, United States: Today, Xlear is filing a Pre-Emergency Use Authorization (Pre-EUA) Request with the U.S. Food and Drug Administration (FDA). The Pre-EUA is a first step in seeking authorization of Xlear Nasal Spray as…
Read More » -

-
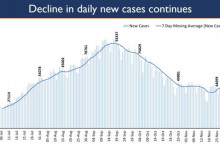
-

-

-

-

-
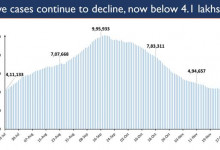
-

-
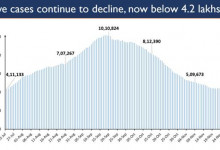
-

-

-

-
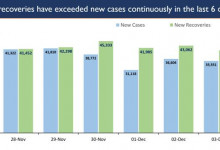
-
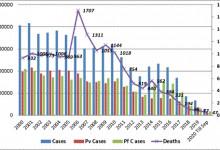
-
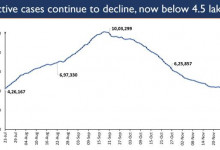
-

-
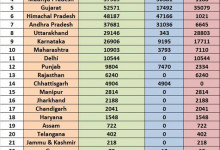
-

-

-
-

-

-

-

-

-
-

-

-